Periodic table with atomic number4/1/2023  Working with elements from aluminum (which has an atomic number thirteen) to gold (seventy nine), he was able to show that the frequency of these transitions increased with each element studied. The atomic number can be used to uniquely identify ordinary chemical elements. For ordinary nuclei, this is equal to the proton number ( np) or the number of protons found in the nucleus of every atom of that element. Moseley was able to confirm these two hypotheses through experimentation, measuring the wavelengths of photon transitions of various elements while they were inside an x-ray tube. The atomic number or nuclear charge number (symbol Z) of a chemical element is the charge number of an atomic nucleus. 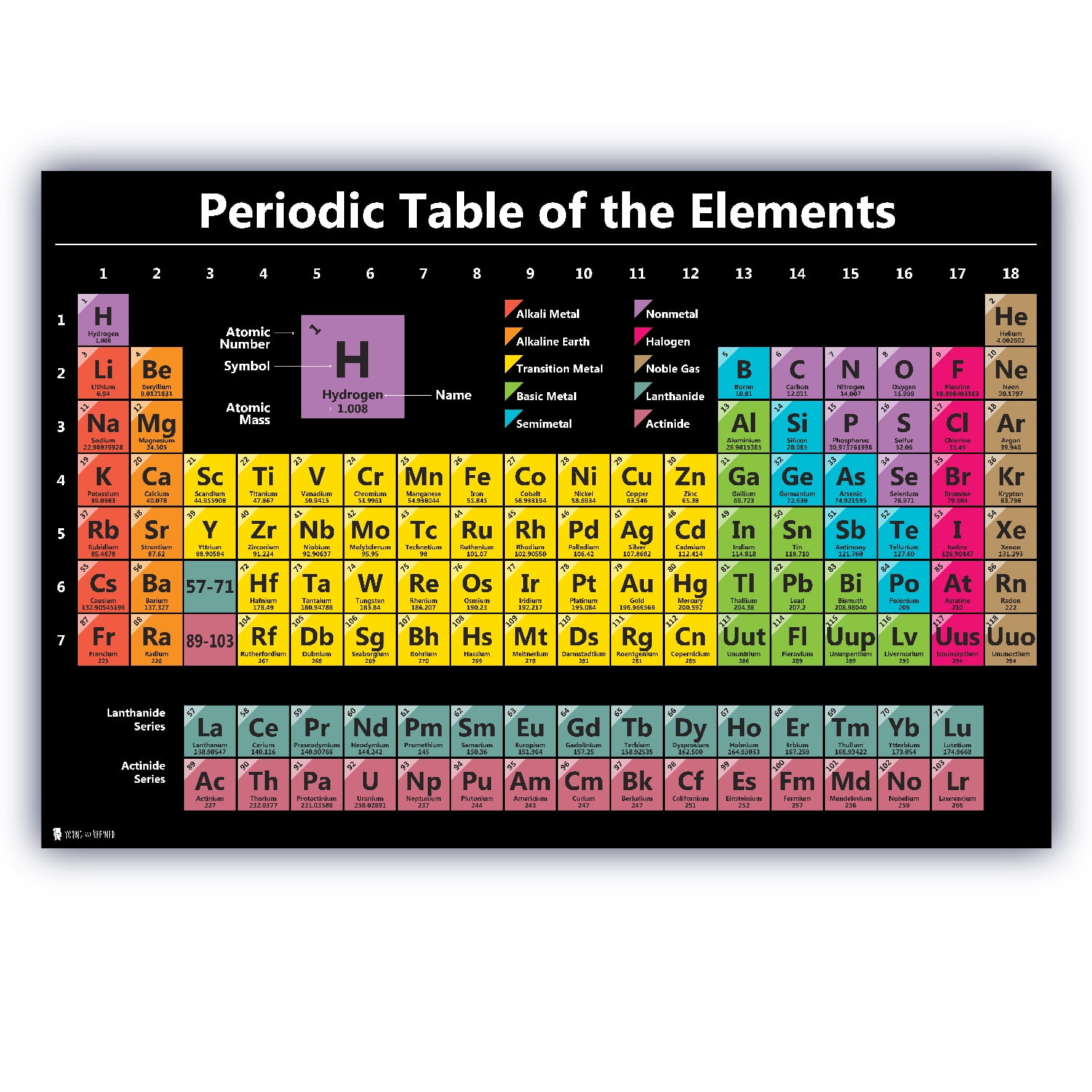
The Bohr model of the atom had the central charge contained in its core, with its electrons circulating it in orbit, much like how the planet in the solar system orbit the sun. Two years later, Henry Moseley and Niels Bohr made further contributions that helped to confirm this. Antonius van den Broek added to this by formerly suggesting that the central charge and number of electrons were equal. This central charge would be roughly equal to half of the atoms total atomic weight. The heaviest element in nature is uranium, which has 92. An elements atomic number is the number of protons it contains in its nucleus. Visualize trends, 3D orbitals, isotopes, and mix compounds. Meet 115, the Newest Element on the Periodic Table. It was he who first suggested the model for an atom where the majority of its mass and positive charge was contained in a core. Interactive periodic table showing names, electrons, and oxidation states. The atomic number of an element never changes, meaning that the number of protons in the nucleus of every atom in an element is always the same.Īrranging elements based on their atomic weight began with Ernest Rutherford in 1911. Oxygen atoms contain 8 protons and have an atomic number of 8, and so on. All carbon atoms contain six protons and therefore have an atomic number of 6. For example, Hydrogen atoms, which have one proton in their nucleuses, are given an atomic number of one. This fact has key implications for the building up of the periodic table of elements.Ever wonder why the periodic table of elements is organized the way it is? Why, for example, does Hydrogen come first? And just what are these numbers that are used to sort them all? They are known as the element’s atomic number, and in the periodic table of elements, the atomic number of an element is the same as the number of protons contained within its nucleus. The ordering of the electrons in the ground state of multielectron atoms, starts with the lowest energy state (ground state) and moves progressively from there up the energy scale until each of the atom’s electrons has been assigned a unique set of quantum numbers. It is the Pauli exclusion principle that requires the electrons in an atom to occupy different energy levels instead of them all condensing in the ground state. In the periodic table, the elements are listed in order of increasing atomic number Z. For instance, Mendeleevs 1869 periodic table. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The atomic number was simply an ordinal number assigned to each element, and it had no physical significance. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. See also: Atomic Number – Does it conserve in a nuclear reaction? Atomic Number and Chemical PropertiesĮvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. 
Each element has its own atomic number, which represents the number of protons in one atom of the element. It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. As you move across the table, the number of protons and the atomic mass of each element increases. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. When atomic number, rather than atomic mass, is used to construct a periodic table, these problems disappear, since an elements chemical properties depend. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
